7. Explain the shape of water molecule with the help of Molecular Orbital Theory, also draw its orbital diagram. (b) What are the gamma rays? Explain their applications.
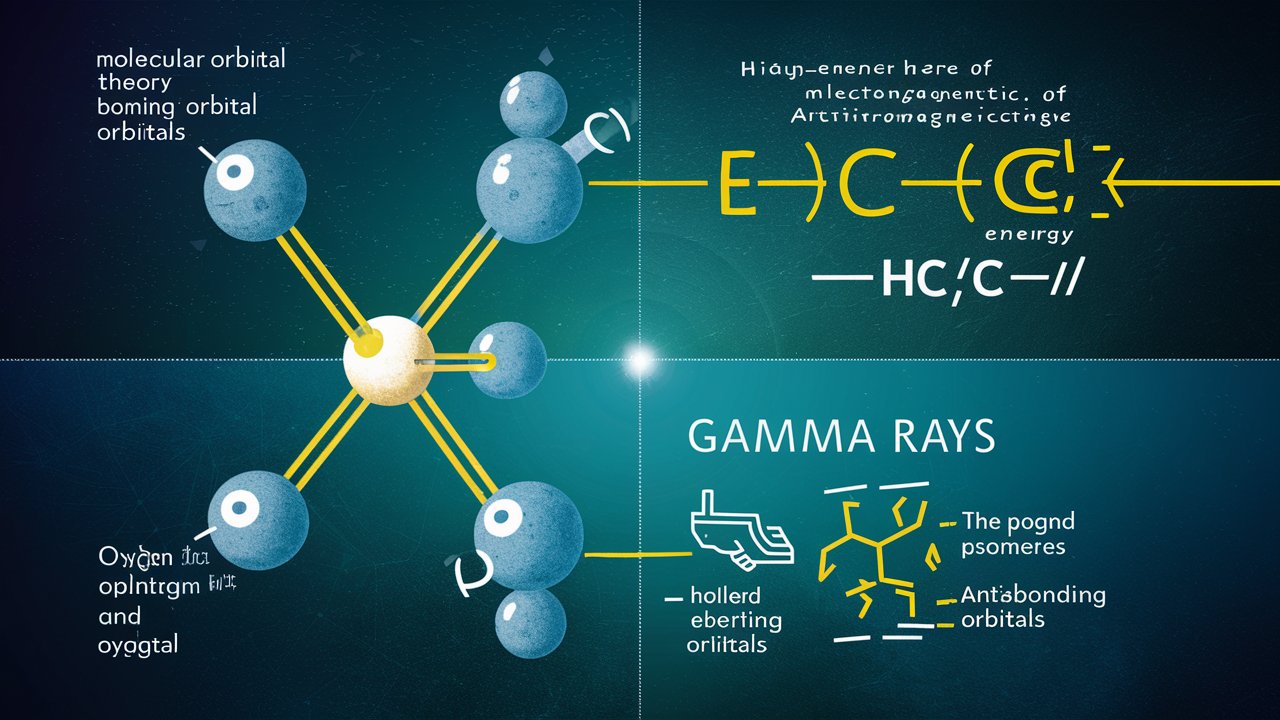
Explain the shape of water molecule with the help of Molecular Orbital Theory, also draw its orbital diagram. (b) What are the gamma rays? Explain their applications.
a) The shape of a water molecule can be explained using Molecular Orbital Theory, which is a quantum mechanical model used to describe the behavior of molecules. According to this theory, the water molecule is composed of two hydrogen atoms and one oxygen atom, and the electrons in these atoms are arranged in molecular orbitals.
The molecular orbitals in a water molecule can be represented using an orbital diagram, which shows the relative energies and spatial arrangements of the orbitals. The diagram for a water molecule shows two bonding orbitals, which hold the two hydrogen atoms to the oxygen atom, and two non-bonding orbitals, which are located on the oxygen atom itself. The non-bonding orbitals are responsible for the bent shape of the water molecule, as they occupy more space than the bonding orbitals and cause the hydrogen atoms to be slightly offset from each other.
b) Gamma rays are a type of electromagnetic radiation, similar to X-rays, but with much higher energy and shorter wavelengths. They are produced by the decay of radioactive atoms and can also be created through nuclear reactions.
Gamma rays have many important applications in medicine, industry, and research. In medicine, they are used for cancer treatment, as they can be directed at cancer cells to kill them without damaging healthy tissue. In industry, they are used to sterilize medical equipment and food products, as they can kill bacteria and other microorganisms. In research, they are used to study the structure of materials and to investigate the behavior of particles at the atomic and subatomic level.
However, gamma rays can also be dangerous, as they can ionize atoms and molecules, causing damage to DNA and other biological molecules. Exposure to high levels of gamma radiation can cause radiation sickness, cancer, and other health problems. Therefore, it is important to use proper safety measures when working with gamma rays and other sources of ionizing radiation.
visit: https://techinsightguru.com/